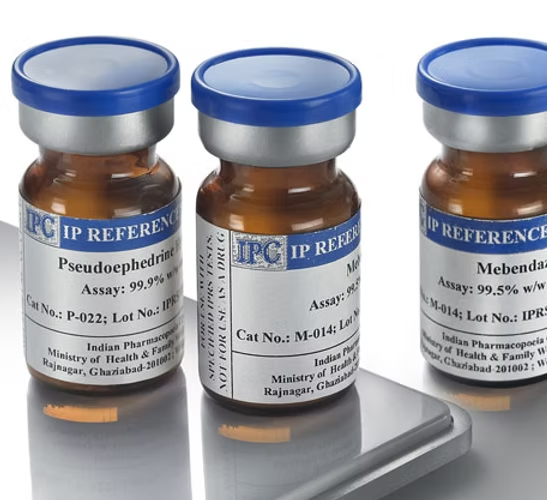
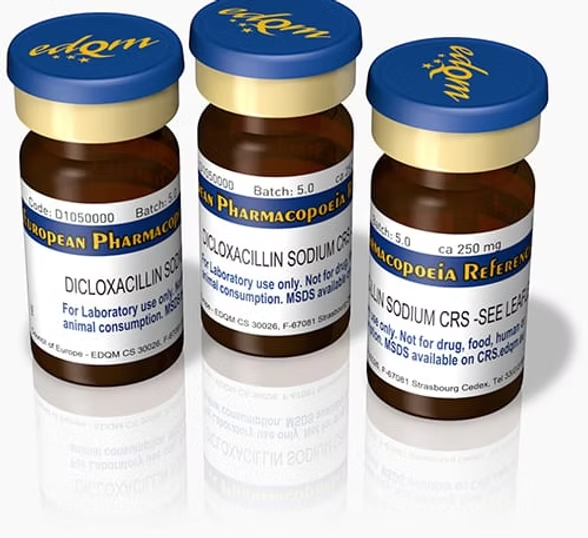
Reference Standard & Impurity

Reference standards are highly purified, well-characterized materials used to verify the identity, strength, quality, and purity of chemical, pharmaceutical, and medicinal substances.
They serve as the benchmark for analytical testing and quality control processes.
Working standards can be prepared using these reference materials. Reference standards may be sourced from official pharmacopeias such as USP (United States Pharmacopeia), EP (European Pharmacopeia), BP (British Pharmacopeia), IP (Indian Pharmacopeia), or developed in-house under strict validation protocols.